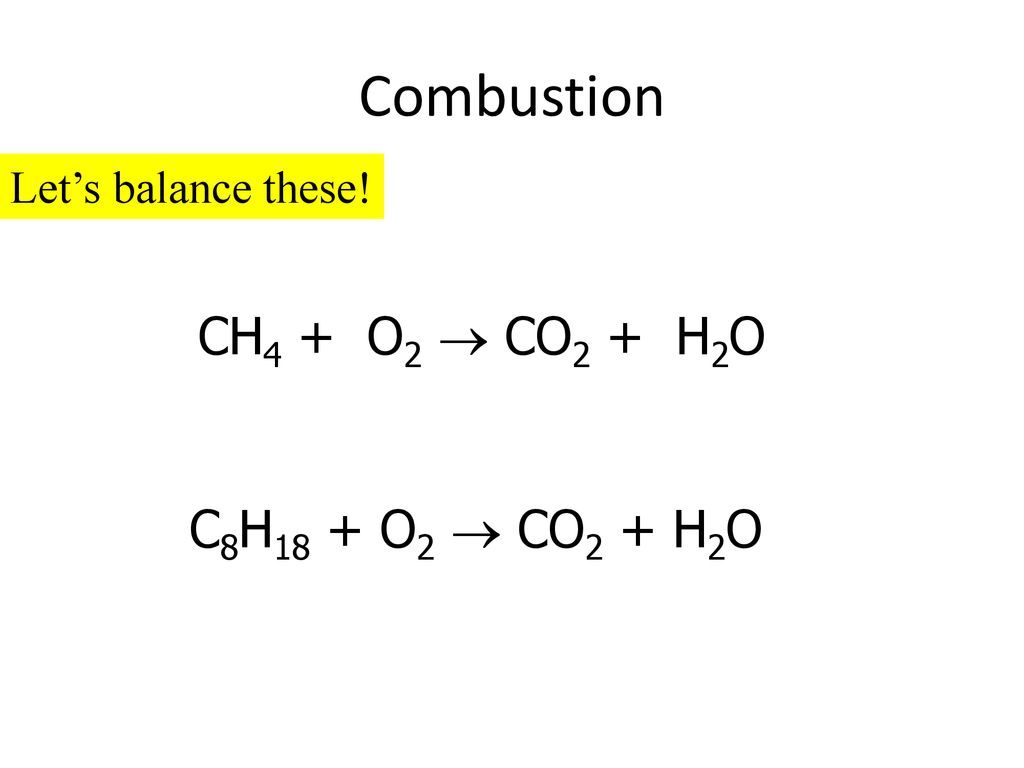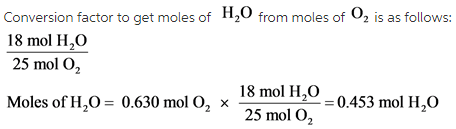
Determine the balanced chemical equation for this reaction. C8H18(g)+O2(g)→ CO2(g)+H2O(g) - Home Work Help - Learn CBSE Forum

How to C8H18+O2=CO2+H2O|Chemical equation C8H18+O2=CO2+H2O|Reaction balance C8H18+O2=CO2+H2O| - YouTube

When octane (C8H18) is burned in air, it chemically reacts with oxygen gas ( O2) to produce carbon dioxide (CO2) and water (H2O). What mass of water is produced by the reaction of
When 10 g of CxHy is burned, 45.00 g of products are formed (CO2 and H2O combined), what is the empirical formula of the fuel (CxHy + O2→ CO2 + H2O)? - Quora
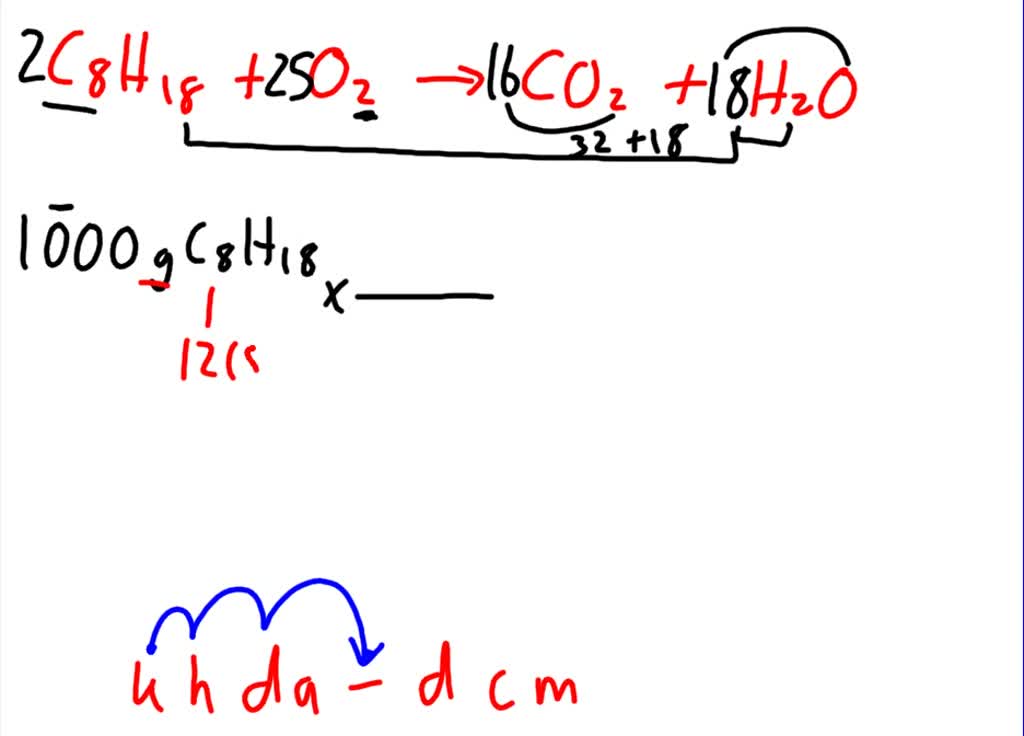
SOLVED:The combustion of gasoline produces carbon dioxide and water. Assume gasoline to be pure octane (C8H18) and calculate the mass (in kg) of carbon dioxide that is added to the atmosphere per

SOLVED: SECTION 3: ANSWER ONE QUESTION FROM THIS SECTION. Balance the following chemical reactions. (2 Marks Each) C8H18 + O2 → CO2 + H2O C9H20 + O2 → CO2 + H2O +