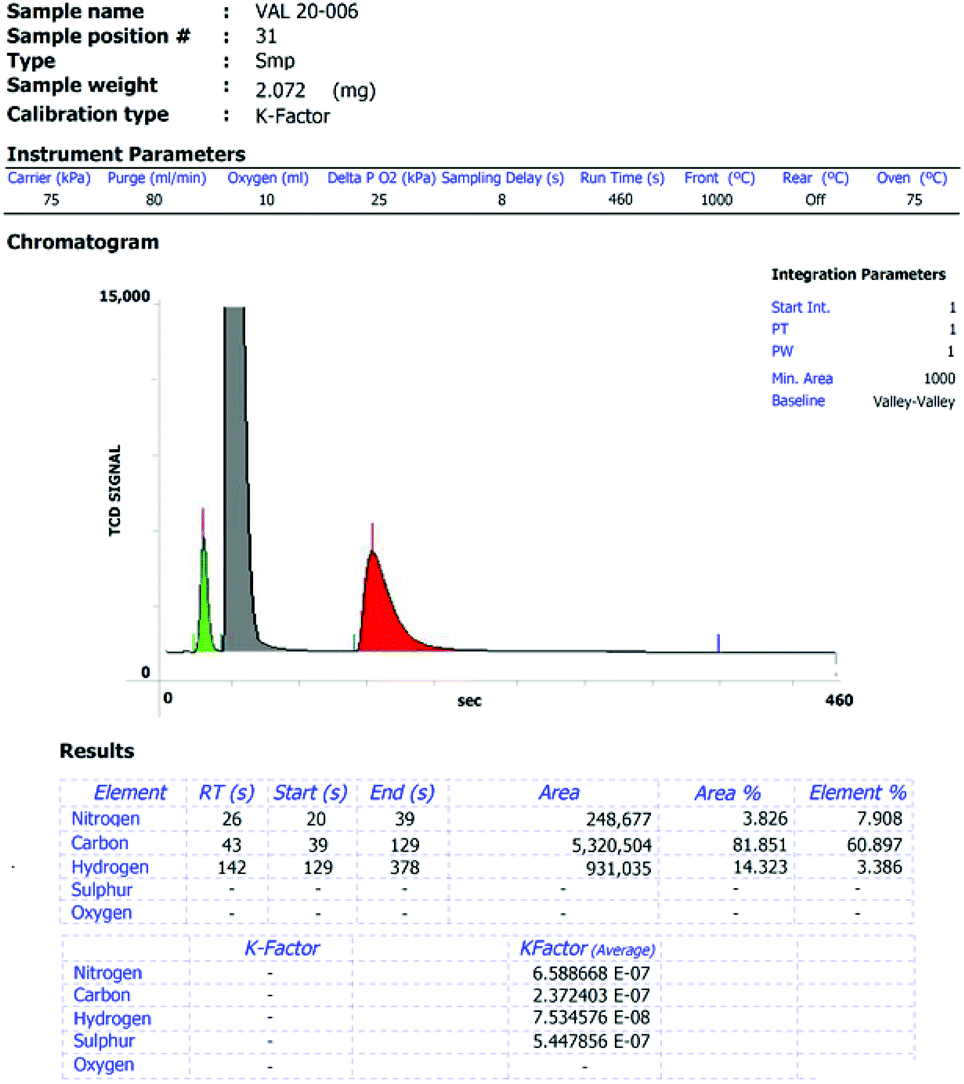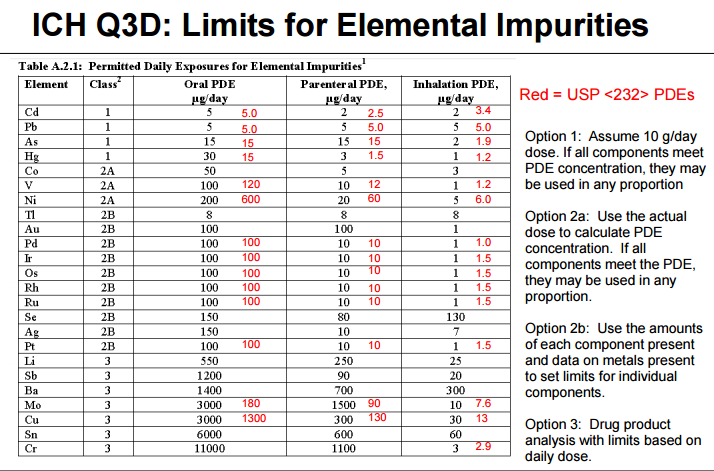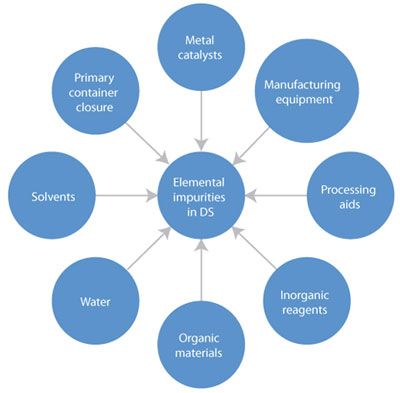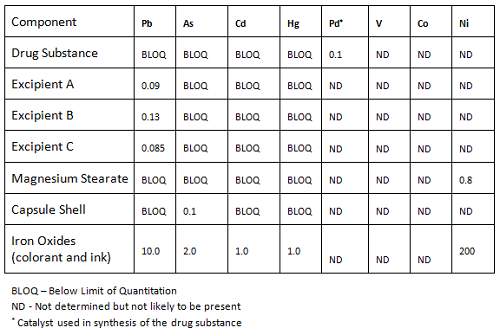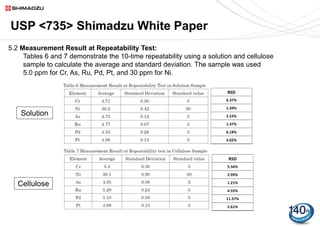
USP 735 as an Alternative to USP 233 for Elemental Impurity Analysis in Pharmaceutical Products | PPT

Elemental Impurities: Implications for Manufacturers of Drug Products, APIs, and Excipients | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology

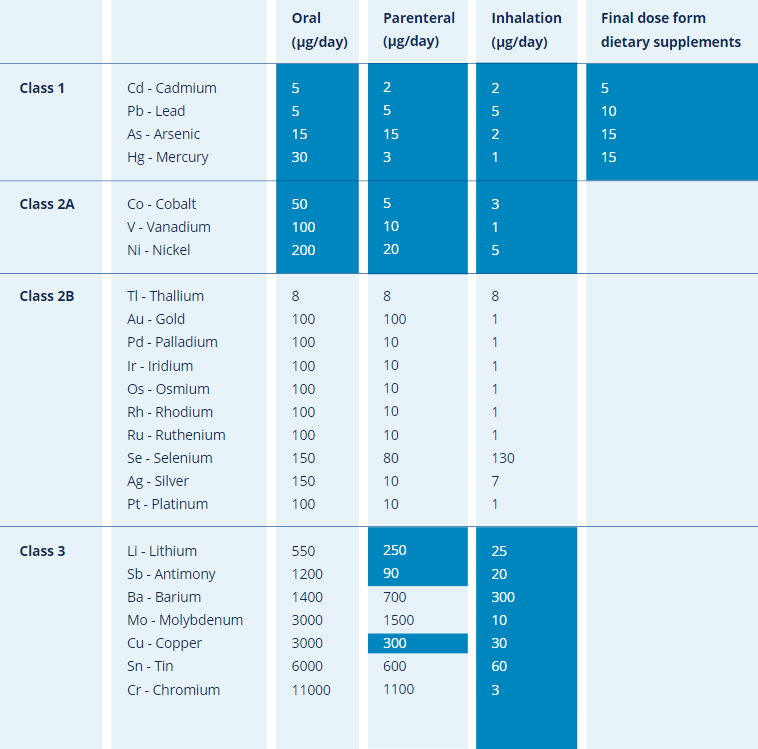
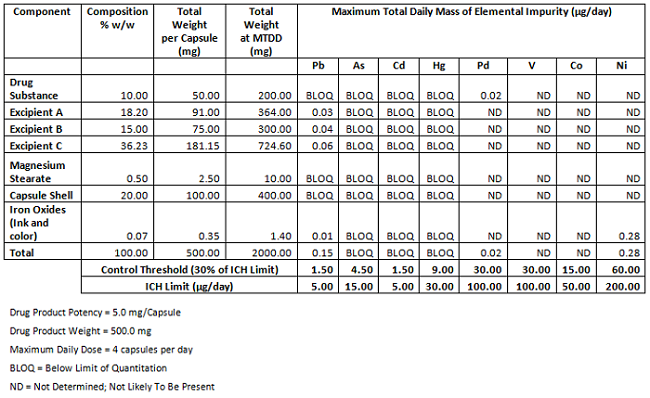
Calculation elemental impurities by Option 2b as per ICH Q3D. | How to calculate elemental impurities by Option 2b as per ICH Q3D (Guideline for Elemental Impurities)? | By Pharma Content | Facebook

Question Video: Determining the Impurity Percentage Given the Mass of the Desired Substance and the Total Mass | Nagwa

An inductively coupled plasma mass spectrometry method for the determination of elemental impurities in calcium carbonate mineral medicine - ScienceDirect

Identification and Characterization of an Isolated Impurity Fraction: Analysis of an Unknown Degradant Found in Quetiapine Fumarate | Waters