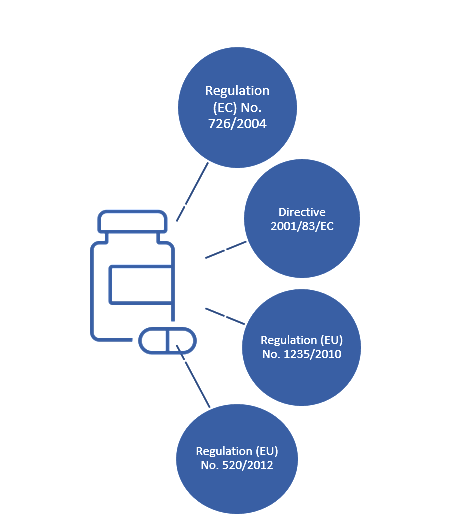
Risk-Based Biologics: CMC Flexibilities in the EU Regulatory System - BioProcess InternationalBioProcess International

Data Safety and Monitoring Boards Should Be Required for Both Early- and Late-Phase Clinical Trials - ScienceDirect

Evaluation of quantitative signal detection in EudraVigilance for orphan drugs: possible risk of false negatives | Semantic Scholar
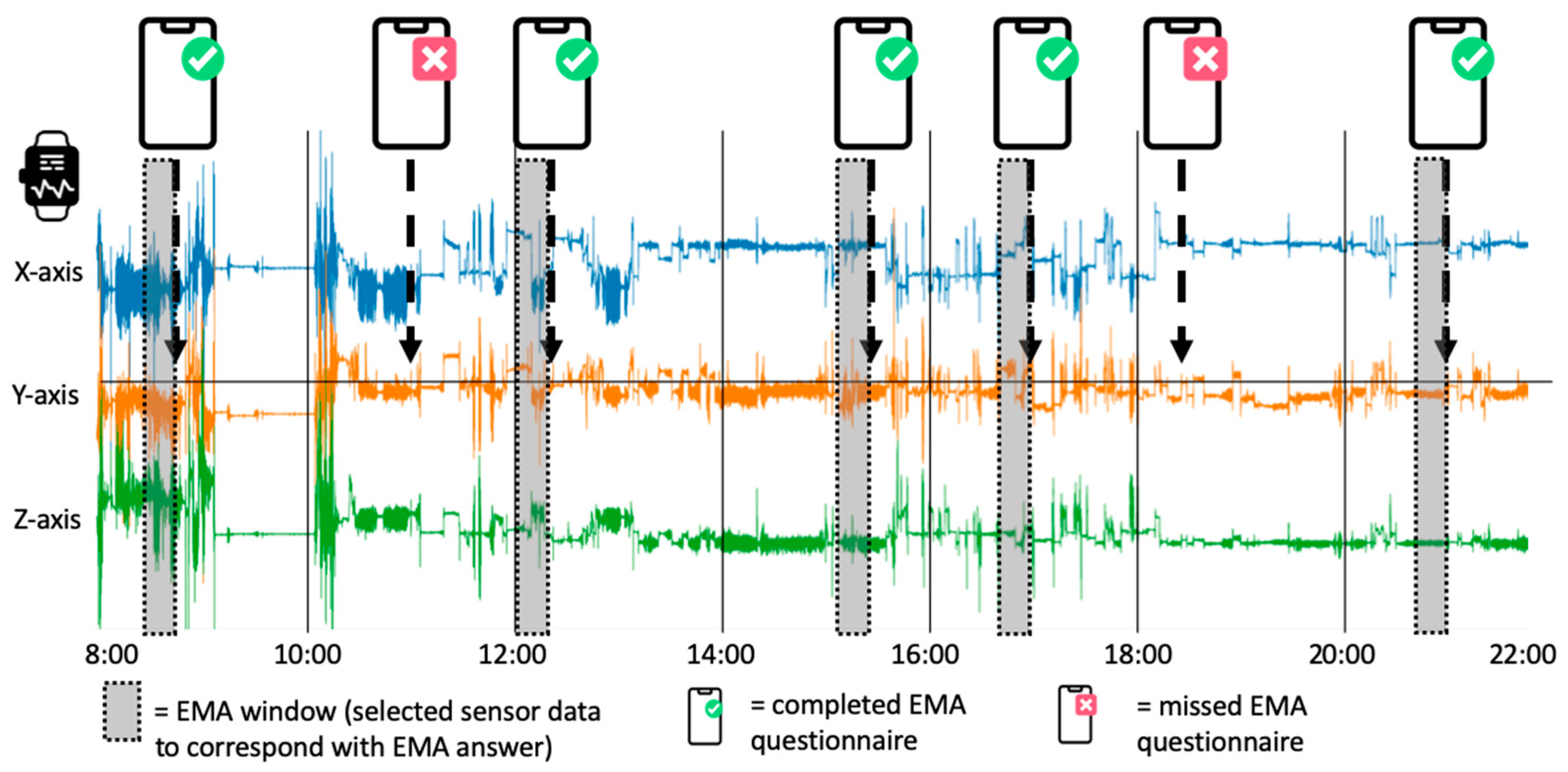
Data | Free Full-Text | A Long-Term, Real-Life Parkinson Monitoring Database Combining Unscripted Objective and Subjective Recordings

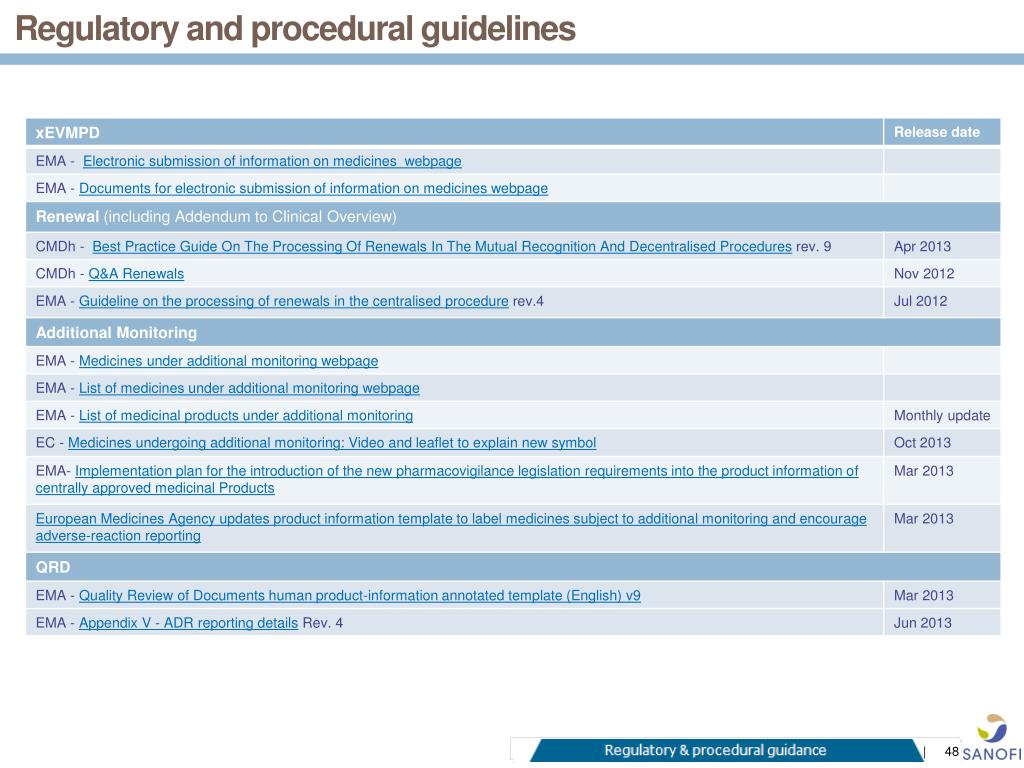
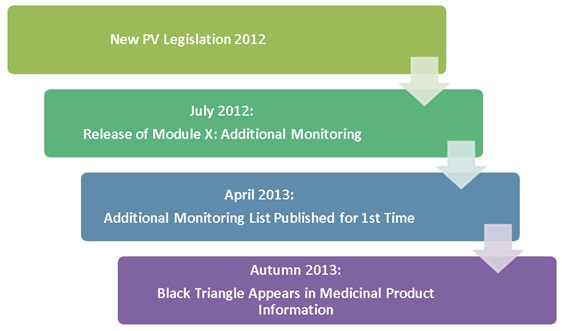
EU Commission Report on National and EMA experience on Medicines subject to Additional Monitoring released today

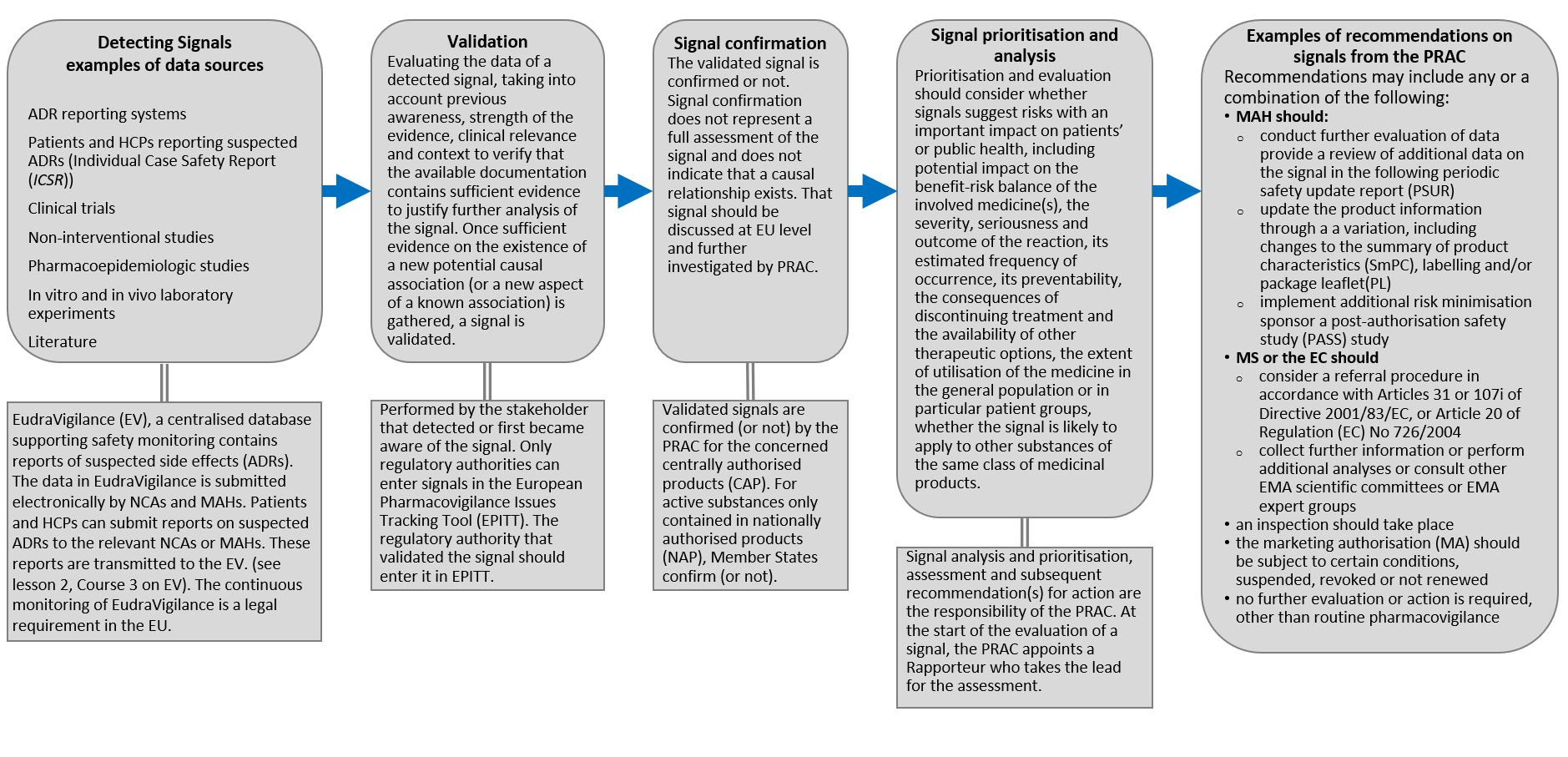
Improving the Safety of Medicines in the European Union: From Signals to Action - Potts - 2020 - Clinical Pharmacology & Therapeutics - Wiley Online Library

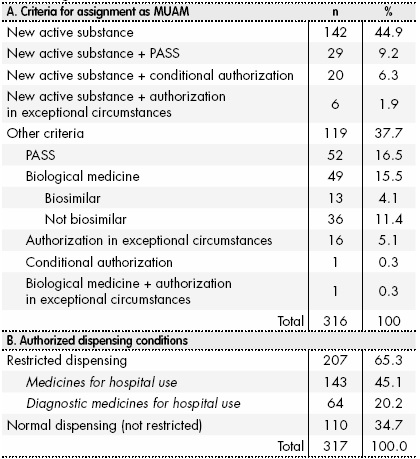
PDF) Does additional monitoring status increase the reporting of adverse drug reaction s ? An interrupted time series analysis of EudraVigilance data
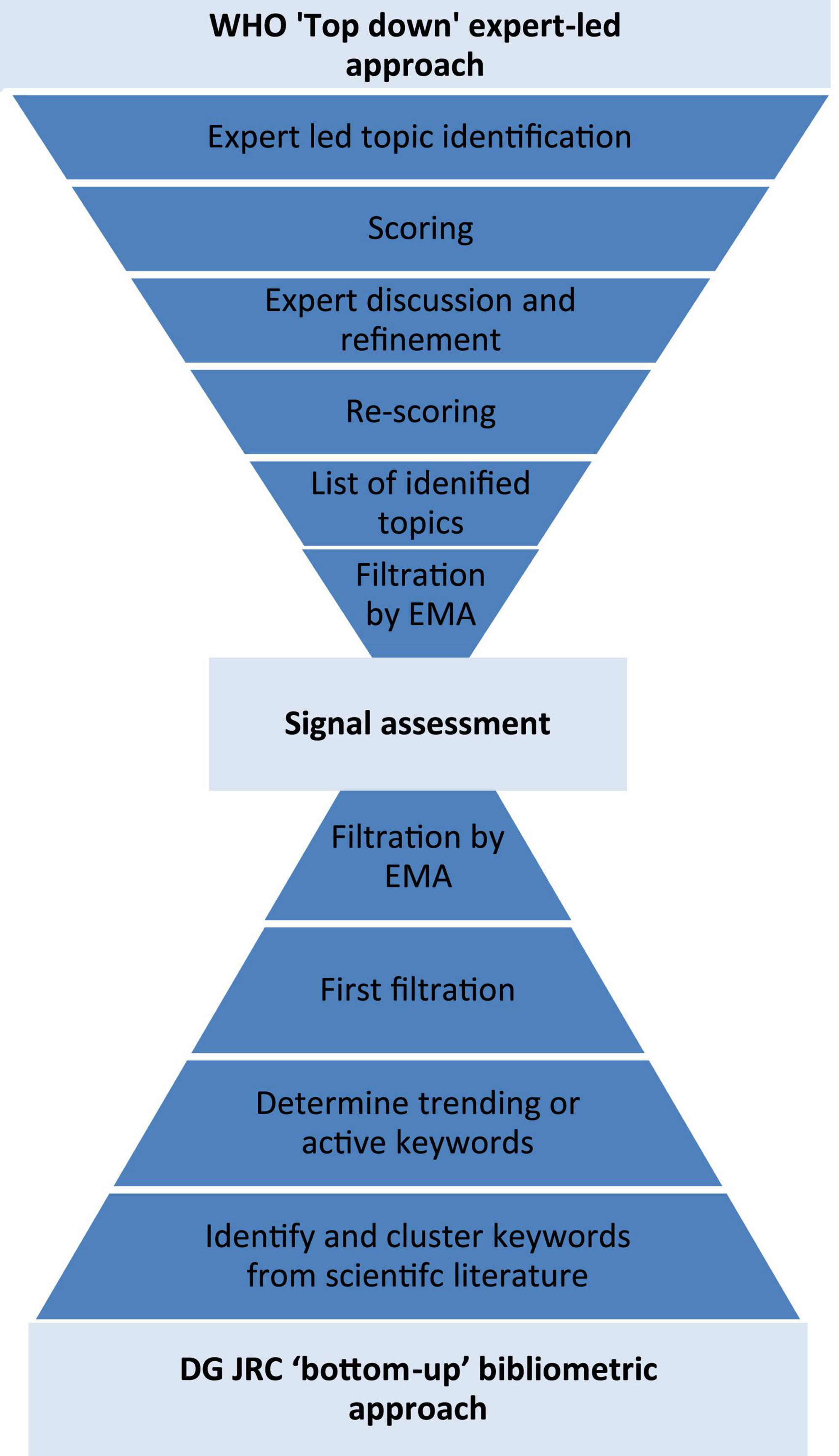
Frontiers | Health horizons: Future trends and technologies from the European Medicines Agency's horizon scanning collaborations

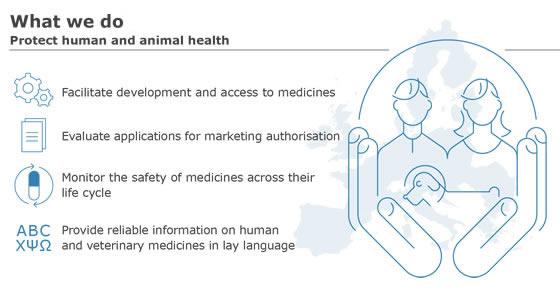
Promoting and Protecting Public Health: How the European Union Pharmacovigilance System Works. - Abstract - Europe PMC