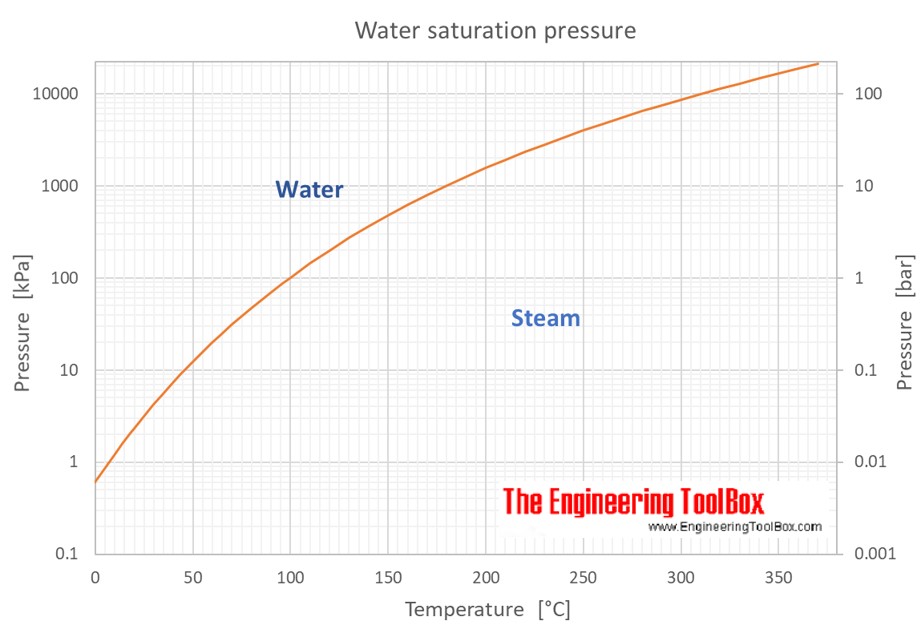
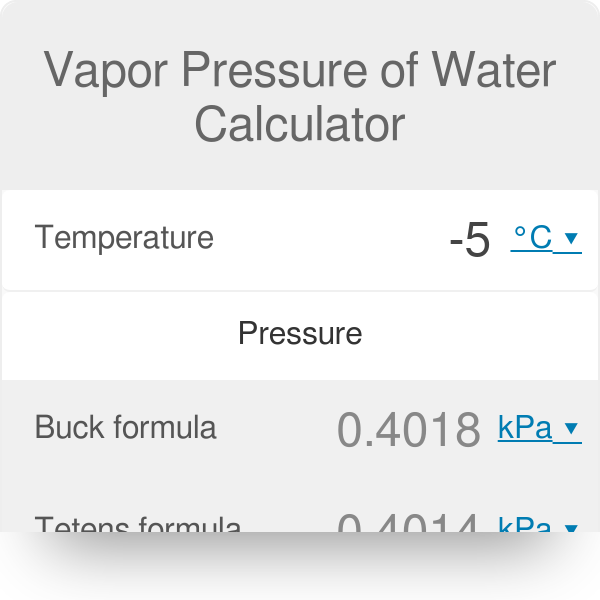
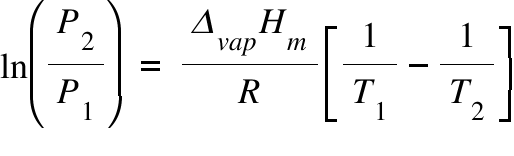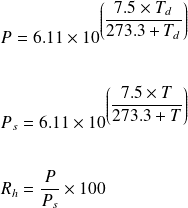A Simple Accurate Formula for Calculating Saturation Vapor Pressure of Water and Ice in: Journal of Applied Meteorology and Climatology Volume 57 Issue 6 (2018)

The vapour pressure of a pure liquid 25°C is 100 mm Hg. Calculate the relative lowering vapour pressure the mole fraction of solvent in solution is 0.8. pº - PS = Xsolute
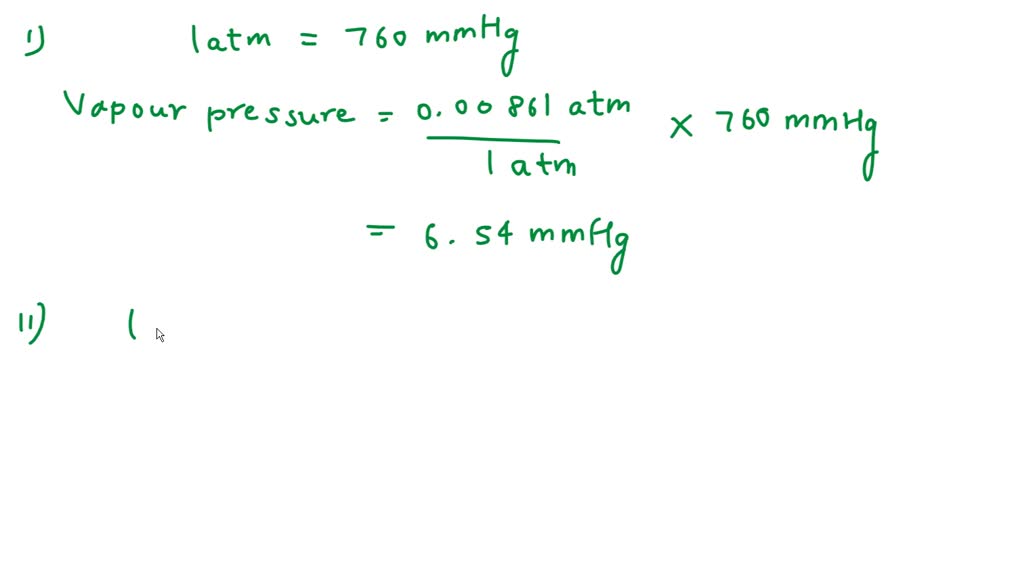
SOLVED: The vapor pressure of water at 5°C is 0.00861 atm. Calculate the vapor pressure in mmHg and torr. Round each of your answers to 3 significant digits. mmHg? torr?
The vapour pressure of pure water is 760 mm at 25^°C . The vapour pressure of solution containing 1(m) solution of glucose will be ??

a) Vapour pressure of benzene is 200 mm of Hg. When 2 gram of a non-volatile solute dissolved in 78 gram benzene. Benzene has vapour pressure of 195 mm of Hg. Calculate
Calculate vapour pressure of 0.1M urea soln. Vapour pressure of water at the given temperature is 20 torr. Assume molality and molality to be equal.